At Meril Life Sciences we strive to improve the quality of human lives by manufacturing innovative solutions that empower healthcare professionals and the medical fraternity. Our manufacturing facilities adhere to cGMP guidelines, are ISO 13845:2003 certified and DCG (I)-approved. Over the years, we have created over 140 products that adhere to the highest possible standards of quality and innovations spread across 5 divisions and are available in more than 100 countries. At Meril, our continual goal is to identify unmet clinical needs and churn out relevant devices in quick turnaround cycles. We strive to be trendsetters in new product introductions. That’s where our dedicated R&D facility comes in, which hosts more than 150 R&D personnel, working in cross functional capacities. These innovators are tasked with creating top of the line products that adhere to each and every safety guideline while still pushing the envelope by creating healthcare solutions that are effective. To understand how this happens let us take a look at some of our hero products from different divisions and see for ourselves just how much effort goes into each and every single one of our products.
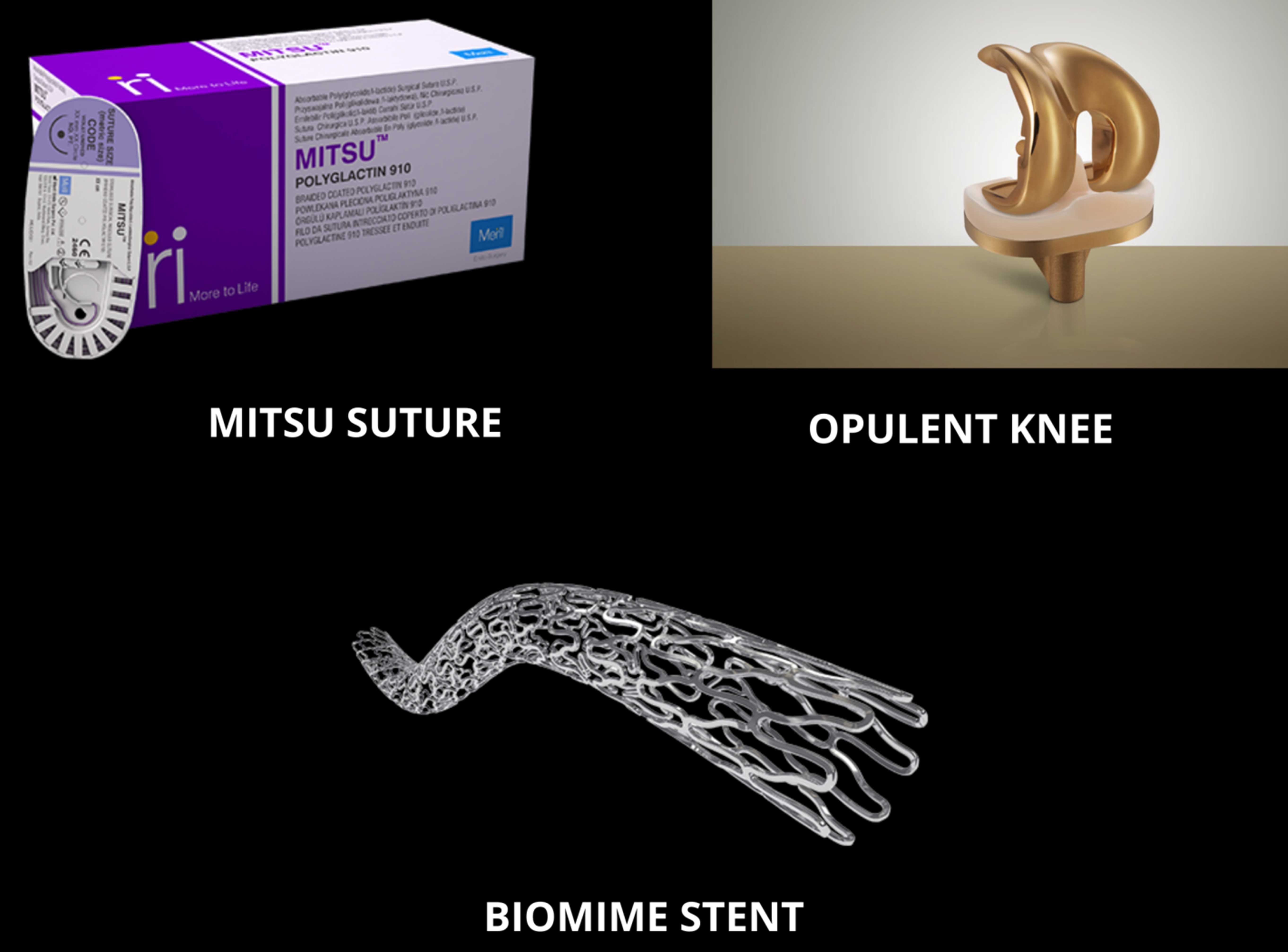
BioMime
Beginning with a medical device that is at the forefront of cardiovascular intervention, we have the BioMime. The BioMime is a Sirolimus Eluting Coronary Stent System with an ultra-thin (65 µm) strut thickness. It is a next-generation SES with a novel & hybrid design consisting of both closed and open cells. It is made from a Biodegradable Polymer with 2µm low thickness which helps facilitate faster healing. The anatomy of a strut in medical devices such as BioMime is a crucial factor in determining how well it can help patients. The strut width variability of the BioMime allows for optimal radial strength & conformability. The BioMime is an assured step towards biomimicry.
Opulent knee
With the help of world-renowned surgeons and a large range of orthopedic devices, Meril Life Sciences has emerged as a frontrunner in the field of orthopedics. The opulent knee is a knee replacement device that revolutionised this industry. For more than 50 years now, cobalt chrome was used to create knee implants. However, to stimulate better performance and patient outcomes, we at Meril Life Sciences made use of a Titanium niobium nitride (TiNbN) coating which is 40% less likely to be worn down than regular cobalt chrome implants. The opulent knee is twice as strong as similar implants in the category; the most biocompatible and non-allergic surface material out there and 8 times harder. With so many positive points working in its favour, the opulent knee system has earned its place as the ideal implant for young and old patients who want to enjoy active, long-term and high-quality performance and provide them with a lasting knee pain remedy.
Mitsu
Meril endosurgery is a one-stop solution for surgeons, providing them with an extensive product portfolio to rely on. Any form of minimally invasive surgery in which a small video endoscope is inserted into the body can be classified as endosurgery. Commonly performed endosurgery procedures include appendectomies, bowel resections, gallbladder surgeries, hernia repairs, splenectomies, and cancer staging operations. Endosurgery does not require a major incision. Small incisions are made in the body through which a flexible endoscope can be inserted allowing internal imaging to be projected. This allows for a robotic procedure which eliminates unwanted motion and improves surgical dexterity. This method allows remote surgery to be performed by skilled surgeons who approach each operation with precision and focus. Meril endosurgery product Mitsu, is a mid-term absorbable braided and coated synthetic suture made of polyglactin. The Mitsu is an absorbable surgical suture with excellent advantages such as gentle knot repositioning, excellent knot security, smooth knot rundown and excellent pliable material that allows for easy handling. Especially Mitsu AB which helps in Antibacterial wound closure and helps in preventing surgical site infection which is very important to take care after any surgery as patients wants to escape and avoid such situation. As a surgical suture, Mitsu degrades by hydrolysis and assures predictable and reliable absorption. It is a testament to just how essential and beneficial surgical sutures can be.
Read more at https://www.merillife.com/blog/medtech/how-our-hero-products-improve-quality-of-life-for-patients